 
Iodine’s properties are much more similar to those of fluorine, chlorine, and bromine than to oxygen, sulfur, and selenium, and the opposite is true for tellurium. He placed tellurium before iodine, for example, even though tellurium is heavier. Sometimes Mendeleev decided the atomic mass must be wrong because the elements seemed to appear in the wrong order. Putting the elements in the correct place on the table still sometimes required correcting their atomic mass. Russian chemist Dmitri Mendeleev is often called "the father of the periodic table." In 1869, he published a version with 63 elements arranged by atomic mass, showing that when the elements were arranged that way, certain characteristics were periodically repeated. The puzzle had both missing and torn pieces, making it even harder to put together. Second, some of the published information about the elements was known to be wrong. First, they knew there were more elements to be discovered and incorporated into the periodic table. Mendeleev's periodic table was first published in the German chemistry journal Zeitschrift fϋr Chemie in 1869. While specific pieces of these early classifications fit well, no system accommodated all of the approximately 60 known elements. They noticed trends and similarities among elements and started dividing them into discrete groups, the best-known of which are Döbereiner's triads and Newlands' octaves. John Newlands of the United Kingdom, Alexandre Béguyer de Chancourtois of France, and Julius Lothar Meyer and Johann Wolfgang Döbereiner of Germany were among the scientists who contributed to developing a periodic table. 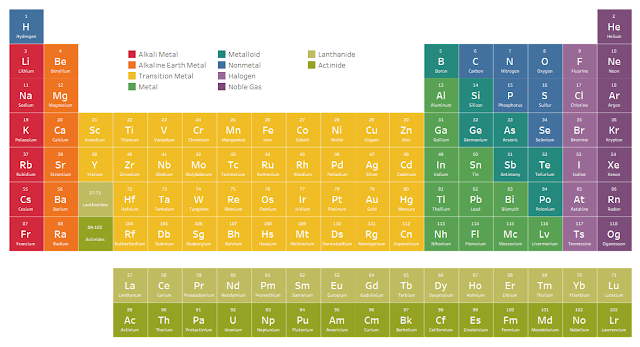
In the 1800s scientists across Europe were working on the same puzzle: making sense of the patterns of behavior observed in chemical elements and developing a systematic way of organizing those elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
